Nature Medicine - AI Section⭐Exploratory3 min read
Key Takeaway:
A new preventive vaccine for Lynch syndrome, a hereditary cancer condition, shows promising safety and immune response in early research, potentially offering future cancer prevention options.
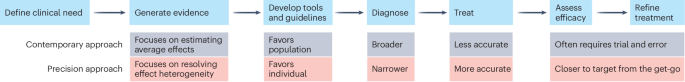
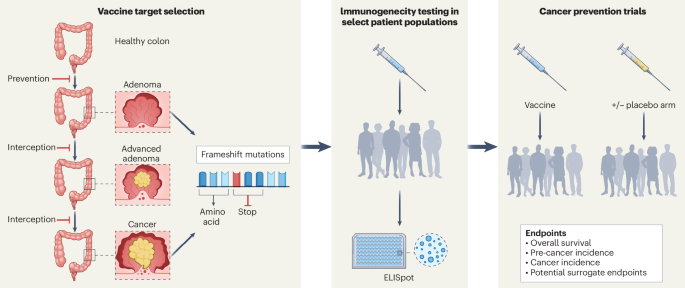
Researchers at the University of California have evaluated the safety and immunogenicity of an 'off-the-shelf' neoantigen vaccine in individuals with Lynch syndrome, a hereditary cancer syndrome, revealing promising results for preventive cancer vaccines. This study is significant as it addresses the growing need for effective prophylactic interventions in hereditary cancer syndromes, which are responsible for a substantial proportion of cancer morbidity and mortality. Lynch syndrome, in particular, predisposes individuals to colorectal and other types of cancer, necessitating novel preventive strategies.
The study employed a phase I clinical trial design involving 30 participants diagnosed with Lynch syndrome. Participants received the neoantigen vaccine, and subsequent immune responses were monitored through blood samples collected at baseline, and at intervals post-vaccination. The primary endpoints were safety, assessed through adverse event reporting, and immunogenicity, measured by T-cell response assays.
Key findings indicated that the vaccine was well tolerated, with no severe adverse events reported. Immunogenicity analysis demonstrated a robust T-cell response in 80% of participants, indicating significant activation of the immune system against neoantigens associated with Lynch syndrome-related tumors. Specifically, post-vaccination, participants exhibited a four-fold increase in neoantigen-specific T-cell activity compared to baseline levels, suggesting the vaccine's potential efficacy in eliciting a targeted immune response.
This research introduces an innovative approach by utilizing a pre-manufactured neoantigen vaccine, which contrasts with the traditionally personalized neoantigen vaccines, thereby simplifying production and potentially reducing costs. However, limitations include the small sample size and the short duration of follow-up, which restrict the ability to assess long-term efficacy and safety comprehensively.
Future directions for this research involve larger-scale clinical trials to validate these findings and evaluate the vaccine's effectiveness in preventing cancer development in Lynch syndrome patients over a longer period. Additionally, exploration of similar vaccine strategies for other hereditary cancer syndromes could be pursued, potentially broadening the impact of this preventive approach.
For Clinicians:
"Phase I trial (n=30) on neoantigen vaccine for Lynch syndrome shows promising immunogenicity and safety. Limited by small sample size and short follow-up. Await larger trials before considering for prophylactic use in hereditary cancer syndromes."
For Everyone Else:
This early research on a preventive cancer vaccine for Lynch syndrome looks promising, but it's not available yet. It may take years. Continue with your current care and consult your doctor for guidance.
Citation:
Nature Medicine - AI Section, 2026. DOI: s41591-026-04248-2 Read article →