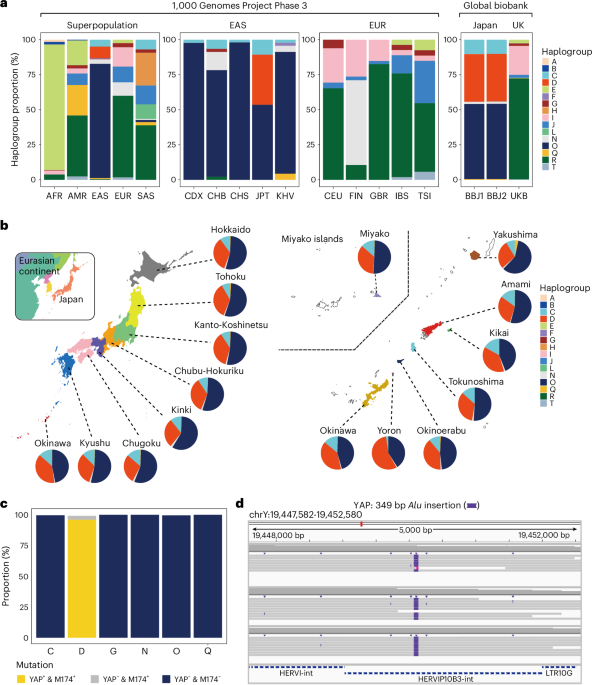
Genetic regulation across germline and somatic variation on the Y chromosome contributes to type 2 diabetes
Key Takeaway:
Research shows that genetic changes on the Y chromosome may influence type 2 diabetes risk differently in East Asian and European men, highlighting a new area for personalized treatment approaches.
For Clinicians:
"Genetic study (n=300,000 males) highlights Y chromosome's role in T2D risk, with differential effects in East Asians vs. Europeans. Early-phase research; clinical application premature. Consider genetic factors in T2D risk assessment cautiously."
For Everyone Else:
Early research suggests the Y chromosome may affect type 2 diabetes risk. It's not ready for clinical use yet. Keep following your current treatment plan and consult your doctor for personalized advice.
Citation:
Nature Medicine - AI Section, 2026. Read article →
